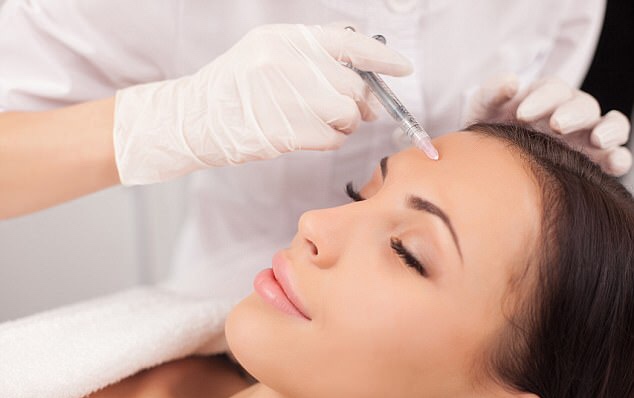
Late last year Tracey Bell told LBN that many people were putting themselves at risk of scarring and lethal complications by paying for Botox and lip filler treatment from poorly-trained practitioners. Tony McDonough reports

Liverpool aesthetics clinic entrepreneur Tracey Bell has welcomed new stricter regulations on the use of dermal fillers – but added people still needed to be wary of unscrupulous practitioners.
Speaking to LBN late last year Dr Bell, who runs private dental and aesthetic clinic in Liverpool and the Isle of Man, said people were putting themselves at risk by paying for Botox and lip filler treatment from poorly-trained practitioners.
She has seen a growing number of young women who’d been left with scarring and potentially fatal complications as a result of facial fillers.
EU regulations
Now, under new EU regulations for medical devices (MDR), dermal fillers will be subject to the same safety checks as medical devices by May 2020 (and this change will still apply post-Brexit).
Dr Bell said: “In this country, we’ve been treating what is a medical product in the US as a beauty treatment, and if something goes wrong, there’s no comeback because there’s no regulation. That’s what a lot of people don’t realise.
“It’s about time British consumers were given the same level of protection they’d have automatically in the States.
“Dermal fillers are a medical treatment and should only be offered in a medical environment, where any complications that occur can be swiftly dealt with and ongoing care can be given.”

Growing demand
Demand for dermal fillers, which are used to add volume to lips and fill wrinkles, has been growing year-on-year in the UK, but until the new regulations were announced this month, they were only subject to the same level of regulation as ballpoint pens and toothbrushes.
However, the EU’s MDR (2017/745) now expressly classifies dermal fillers, even those without a medical purpose, as medical devices, although this will not mean that dermal fillers become prescription only.
Dermal fillers currently placed on the UK market with a medical purpose are classified as medical devices and require a CE mark. There are, however, dermal fillers that are marketed for aesthetic use only that are not classified as medical devices as the manufacturer is not making a medical claim.
Caution needed
Despite the change in regulation Dr Bell says consumers should still be on their guard and urges people considering such treatment to do as much prior research as they can and find out as much as they can about the products being used and the qualifications held by the person who’ll be administering them.
Before the new regulations to come into full force in the UK, Dr Bell will continue to offer only fillers that are FDA approved in her clinics.
